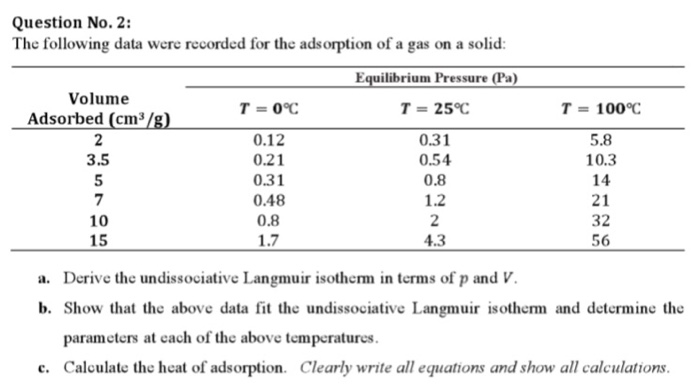
van't Hoff's equation gives the quantitative relation between change in value of K with change in temperature.

PDF) Van't Hoff equation-equilibrium How much Van't Hoff equation -effect on temperature | Kajal Panda - Academia.edu

van't Hoff plot of the equilibrium plateau pressures of the absorption... | Download Scientific Diagram

Plot of Van , t Hoff equation for the adsorption of Cr(VI ), 20, 30, 40... | Download Scientific Diagram
Thermodynamic study of the adsorption of acridinium derivatives on the clay surface - RSC Advances (RSC Publishing) DOI:10.1039/D0RA03158E
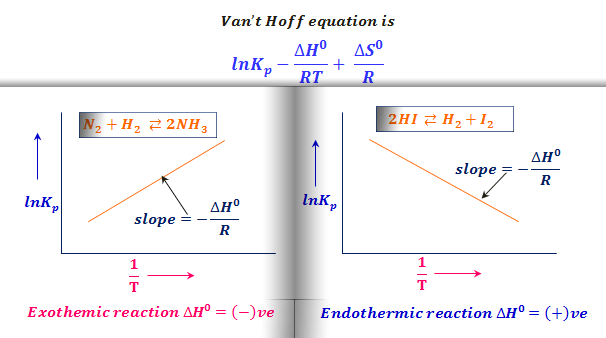
Van't Hoff equation-equilibrium. How much Van't Hoff equation — effect… | by Chemistry Topics | Medium

PPT - Characteristic functions . Thermodynamics of chemical equilibrium PowerPoint Presentation - ID:4206149